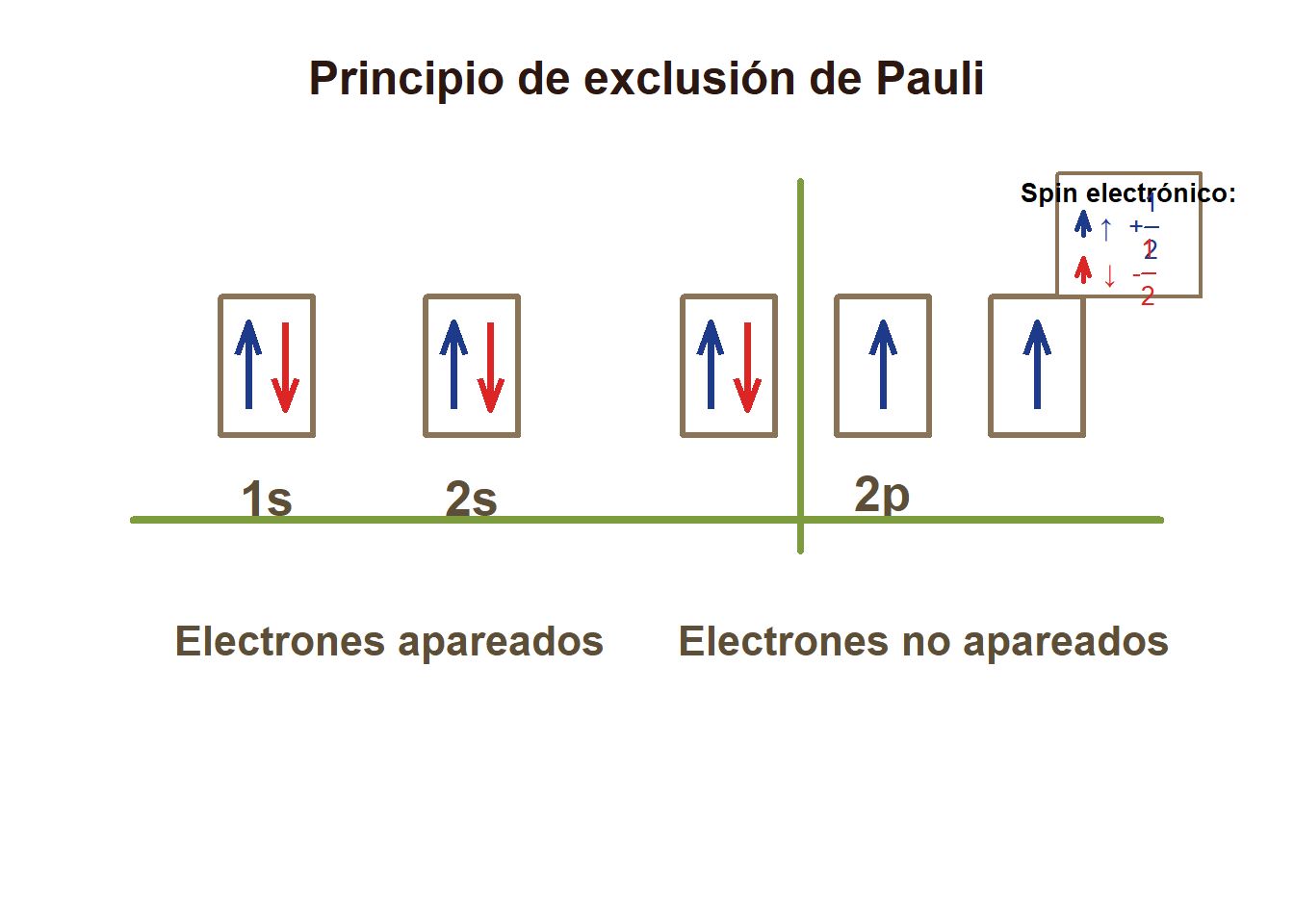
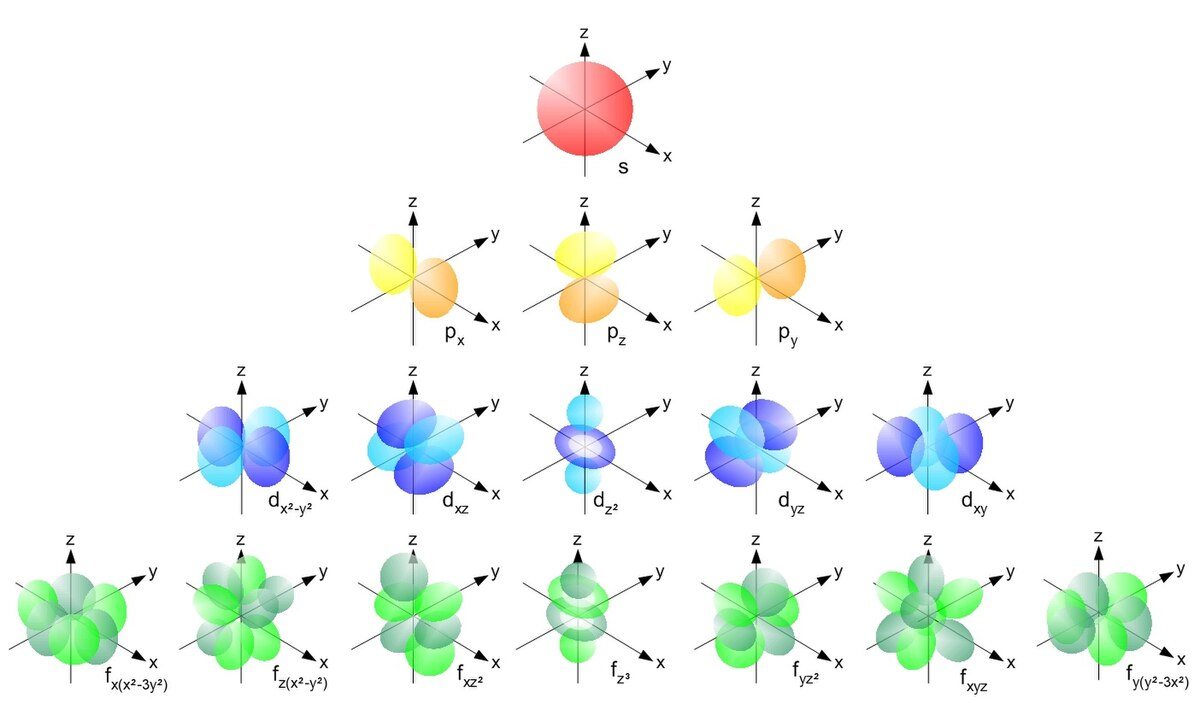
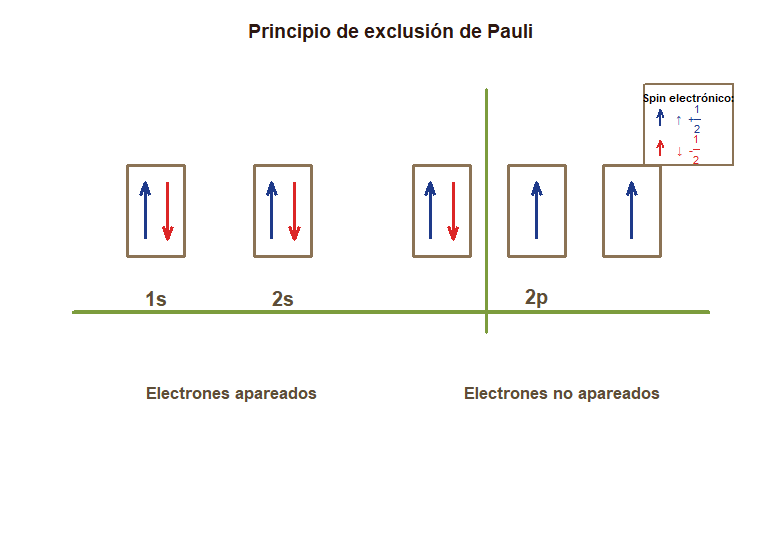
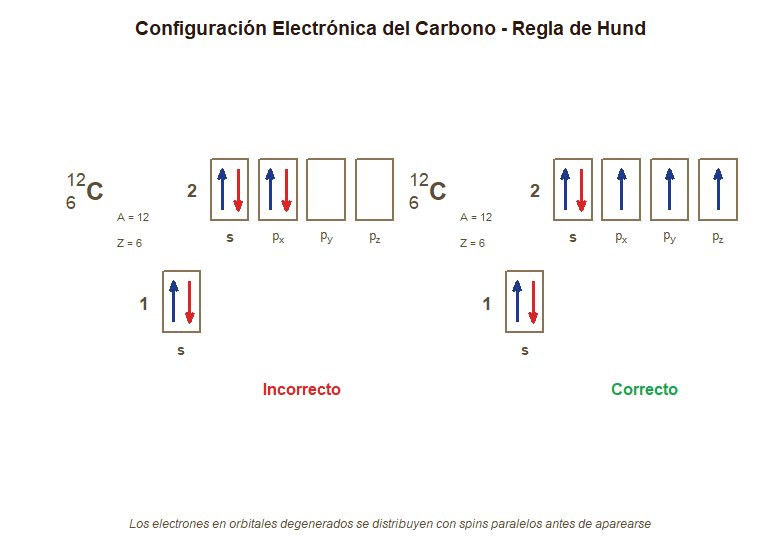
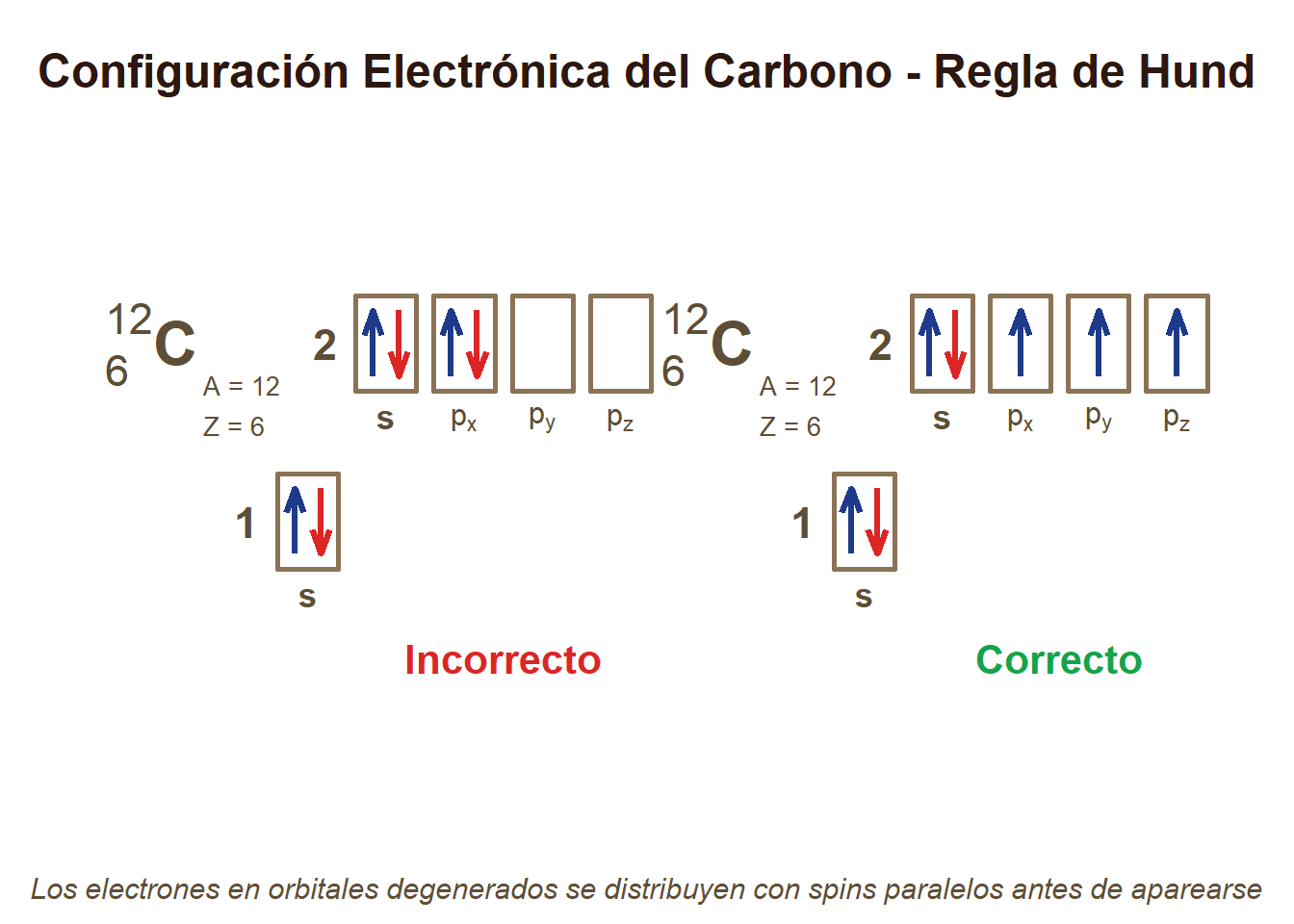
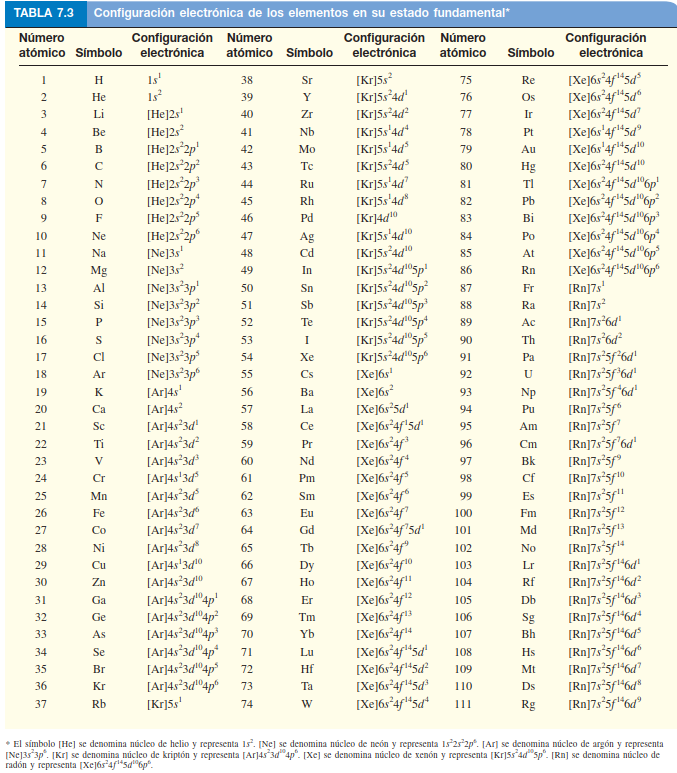
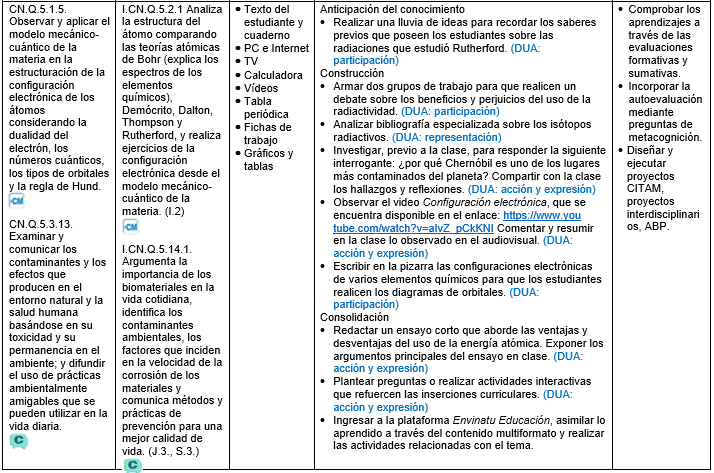
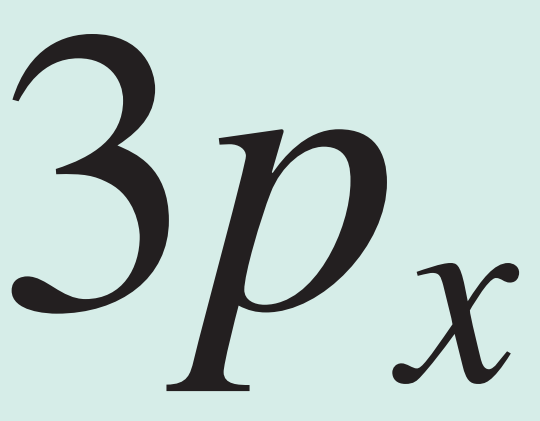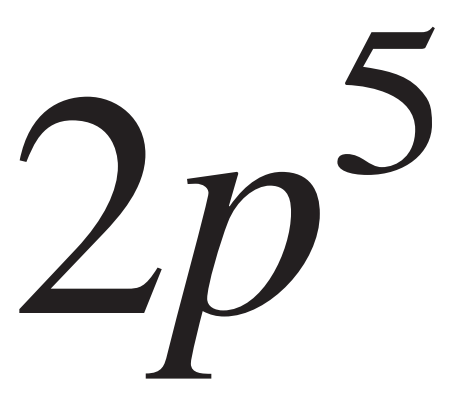# Diagrama de configuración electrónica del Carbono - Regla de Hund
par(mar = c(2, 1, 3, 1), family = "sans", bg = "white")
plot(NULL, xlim = c(0, 13), ylim = c(0, 5),
xlab = "", ylab = "", axes = FALSE, asp = NA)
# Función para dibujar orbitales
dibujar_orbital <- function(x, y, ancho = 0.7, alto = 0.7, col_borde = "#8B7355", lwd_val = 2.5) {
rect(x - ancho/2, y - alto/2, x + ancho/2, y + alto/2,
border = col_borde, lwd = lwd_val, col = "white")
}
# Función para dibujar electrones
dibujar_electron <- function(x, y, direccion = "up", col_flecha = "#2E4057") {
if (direccion == "up") {
arrows(x, y - 0.24, x, y + 0.24,
length = 0.14, lwd = 3, col = col_flecha, angle = 20, lend = 1)
} else if (direccion == "down") {
arrows(x, y + 0.24, x, y - 0.24,
length = 0.14, lwd = 3, col = col_flecha, angle = 20, lend = 1)
}
}
# =============================
# CONFIGURACIÓN INCORRECTA
# =============================
# Símbolo del Carbono
text(0.8, 3.5, expression(bold(""[6]^{12}*C)), cex = 2.0, font = 2, col = "#5D4E37")
text(1.4, 3.2, "A = 12", cex = 0.85, adj = 0, col = "#5D4E37")
text(1.4, 2.9, "Z = 6", cex = 0.85, adj = 0, col = "#5D4E37")
# NIVEL 2
x_base_inc <- 3.5
y_nivel2_inc <- 3.5
text(x_base_inc - 0.7, y_nivel2_inc, "2", cex = 1.4, font = 2, col = "#5D4E37")
# Orbital 2s con electrones apareados
dibujar_orbital(x_base_inc, y_nivel2_inc)
dibujar_electron(x_base_inc - 0.15, y_nivel2_inc, "up", "#1E3A8A")
dibujar_electron(x_base_inc + 0.15, y_nivel2_inc, "down", "#DC2626")
# Orbitales 2p
x_2p_1 <- x_base_inc + 0.9
x_2p_2 <- x_2p_1 + 0.9
x_2p_3 <- x_2p_2 + 0.9
# Primer 2p con electrones apareados (INCORRECTO)
dibujar_orbital(x_2p_1, y_nivel2_inc)
dibujar_electron(x_2p_1 - 0.15, y_nivel2_inc, "up", "#1E3A8A")
dibujar_electron(x_2p_1 + 0.15, y_nivel2_inc, "down", "#DC2626")
# Segundo 2p vacío
dibujar_orbital(x_2p_2, y_nivel2_inc)
# Tercer 2p vacío
dibujar_orbital(x_2p_3, y_nivel2_inc)
# Etiquetas de orbitales 2p
text(x_2p_1, y_nivel2_inc - 0.55, expression(p[x]), cex = 1.0, col = "#5D4E37")
text(x_2p_2, y_nivel2_inc - 0.55, expression(p[y]), cex = 1.0, col = "#5D4E37")
text(x_2p_3, y_nivel2_inc - 0.55, expression(p[z]), cex = 1.0, col = "#5D4E37")
# NIVEL 1 (en columna separada a la izquierda)
x_1s_inc <- 2.6
y_nivel1_inc <- 2.2
text(x_1s_inc - 0.7, y_nivel1_inc, "1", cex = 1.4, font = 2, col = "#5D4E37")
# Orbital 1s con electrones apareados
dibujar_orbital(x_1s_inc, y_nivel1_inc)
dibujar_electron(x_1s_inc - 0.15, y_nivel1_inc, "up", "#1E3A8A")
dibujar_electron(x_1s_inc + 0.15, y_nivel1_inc, "down", "#DC2626")
# Etiqueta s
text(x_1s_inc, y_nivel1_inc - 0.55, expression(bold("s")), cex = 1.1, font = 2, col = "#5D4E37")
text(x_base_inc, y_nivel2_inc - 0.55, expression(bold("s")), cex = 1.1, font = 2, col = "#5D4E37")
# Etiqueta INCORRECTO
text(x_base_inc + 1.35, 1.2, "Incorrecto", cex = 1.3, font = 2, col = "#DC2626")
# =============================
# CONFIGURACIÓN CORRECTA
# =============================
# Símbolo del Carbono
text(7.2, 3.5, expression(bold(""[6]^{12}*C)), cex = 2.0, font = 2, col = "#5D4E37")
text(7.8, 3.2, "A = 12", cex = 0.85, adj = 0, col = "#5D4E37")
text(7.8, 2.9, "Z = 6", cex = 0.85, adj = 0, col = "#5D4E37")
# NIVEL 2
x_base_cor <- 9.9
y_nivel2_cor <- 3.5
text(x_base_cor - 0.7, y_nivel2_cor, "2", cex = 1.4, font = 2, col = "#5D4E37")
# Orbital 2s con electrones apareados
dibujar_orbital(x_base_cor, y_nivel2_cor)
dibujar_electron(x_base_cor - 0.15, y_nivel2_cor, "up", "#1E3A8A")
dibujar_electron(x_base_cor + 0.15, y_nivel2_cor, "down", "#DC2626")
# Orbitales 2p
x_2p_cor_1 <- x_base_cor + 0.9
x_2p_cor_2 <- x_2p_cor_1 + 0.9
x_2p_cor_3 <- x_2p_cor_2 + 0.9
# Primer 2p con un electrón desapareado (CORRECTO)
dibujar_orbital(x_2p_cor_1, y_nivel2_cor)
dibujar_electron(x_2p_cor_1, y_nivel2_cor, "up", "#1E3A8A")
# Segundo 2p con un electrón desapareado (CORRECTO)
dibujar_orbital(x_2p_cor_2, y_nivel2_cor)
dibujar_electron(x_2p_cor_2, y_nivel2_cor, "up", "#1E3A8A")
# Tercer 2p con un electrón desapareado (CORRECTO)
dibujar_orbital(x_2p_cor_3, y_nivel2_cor)
dibujar_electron(x_2p_cor_3, y_nivel2_cor, "up", "#1E3A8A")
# Etiquetas de orbitales 2p
text(x_2p_cor_1, y_nivel2_cor - 0.55, expression(p[x]), cex = 1.0, col = "#5D4E37")
text(x_2p_cor_2, y_nivel2_cor - 0.55, expression(p[y]), cex = 1.0, col = "#5D4E37")
text(x_2p_cor_3, y_nivel2_cor - 0.55, expression(p[z]), cex = 1.0, col = "#5D4E37")
# NIVEL 1 (en columna separada a la izquierda)
x_1s_cor <- 9.0
y_nivel1_cor <- 2.2
text(x_1s_cor - 0.7, y_nivel1_cor, "1", cex = 1.4, font = 2, col = "#5D4E37")
# Orbital 1s con electrones apareados
dibujar_orbital(x_1s_cor, y_nivel1_cor)
dibujar_electron(x_1s_cor - 0.15, y_nivel1_cor, "up", "#1E3A8A")
dibujar_electron(x_1s_cor + 0.15, y_nivel1_cor, "down", "#DC2626")
# Etiqueta s
text(x_1s_cor, y_nivel1_cor - 0.55, expression(bold("s")), cex = 1.1, font = 2, col = "#5D4E37")
text(x_base_cor, y_nivel2_cor - 0.55, expression(bold("s")), cex = 1.1, font = 2, col = "#5D4E37")
# Etiqueta CORRECTO
text(x_base_cor + 1.35, 1.2, "Correcto", cex = 1.3, font = 2, col = "#16A34A")
# =============================
# TÍTULO
# =============================
title(main = expression(bold("Configuración Electrónica del Carbono - Regla de Hund")),
cex.main = 1.5, font.main = 2, col.main = "#2C1810", line = 1)
# Nota explicativa
mtext("Los electrones en orbitales degenerados se distribuyen con spins paralelos antes de aparearse",
side = 1, line = 0.5, cex = 0.95, col = "#5D4E37", font = 3)